|
This is a common source of mistakes, as the halogens themselves are coloured. The gain of an electron is reduction, so the halogens are oxidising agents, themselves being reduced and causing the substances they react with to be oxidised. The halogens can all form halide ions (F –, Cl –, Br – and I –) by gaining an electron to become isoelectronic with noble gases. Astatine is a black solidat room temperature.Fluorine is a very pale coloured gas at room temperature.On the basis of the observed properties of chlorine, bromine and iodine we can predict the physical properties of fluorine and astatine (radioactive and decays very quickly, so very low abundance): Note: Iodine is unusual because it SUBLIMES when it is heated (goes straight from solid to vapour without going through a liquid state). These increase with the number of electrons present in the molecule. There are no polar bonds (same atom, so same electronegativity at each end of the bond) and no prospect of hydrogen bonding, so instantaneous dipole-induced dipole (London forces) are the only intermolecular forces present. The trend in boiling points (Fluorine: -188☌, Chlorine: -34.6☌, Bromine: 58.8☌, Iodine: 184☌) and melting points is explained in terms of the increasing strength of the intermolecular forces which hold the halogen molecules to one another. 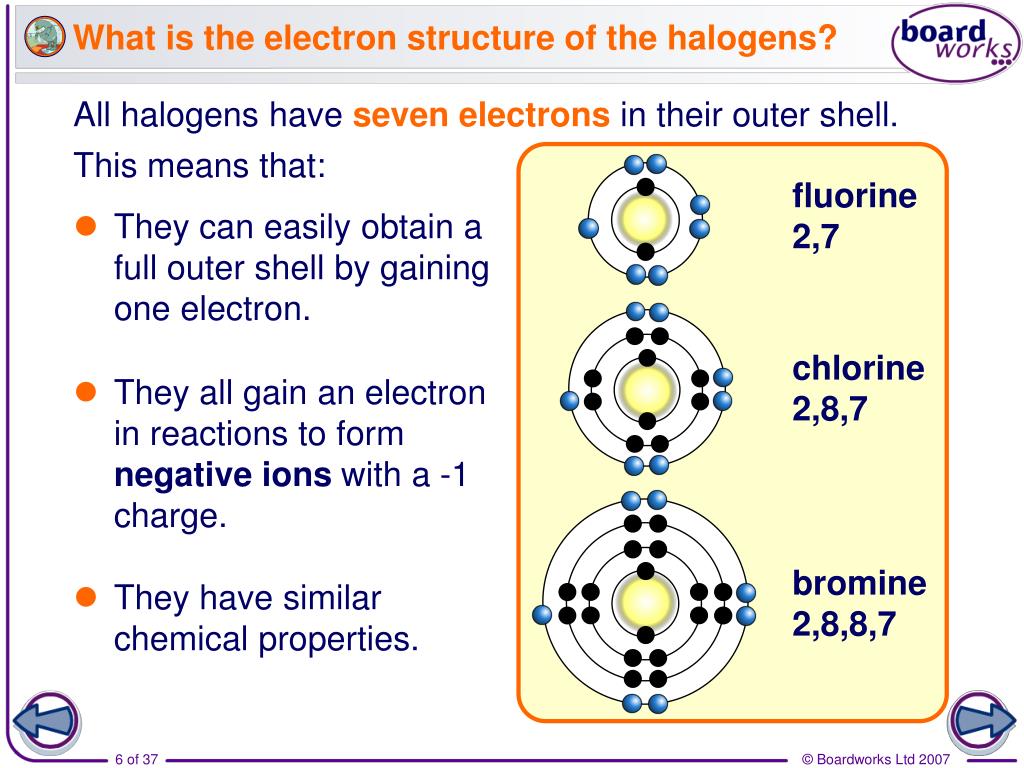
the melting and boiling points increase (gas → liquid → solid at room temp.).Looking at chlorine, bromine and iodine, we can see that down the group: We did not do this at GCSE as it was convenient to have the group number equalling the number of outer-shell electrons, which is why we referred to them as Group 7. * the convention is to number all groups across the periodic table, including the groups of d-block elements. The elements in Group 17 * (the halogens) all exist as diatomic molecules, containing a single covalent bond.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
